Conversions
Before you can solve gas law problems, you will need conversion factors.
Pressure: 1 atm = 760mmHg = 760 torr = 101.3 kPa
Volume: Expressed in cubic meter (cm^3), and most commonly in Liters.
Temperature: 0 degrees Celsius is equivalent to 273 Kelvin.
Pressure: 1 atm = 760mmHg = 760 torr = 101.3 kPa
Volume: Expressed in cubic meter (cm^3), and most commonly in Liters.
Temperature: 0 degrees Celsius is equivalent to 273 Kelvin.
Boyle's Law

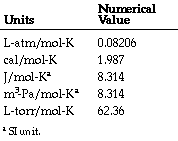
Boyle's Law was discovered by Charles Boyle, who observed the relationship between Pressure and Volume. These are indirectly related, meaning as one increases, the other will decrease, and when one decreases, the other will consequently increase.
Below are a video and an animation for Boyle's Law:
http://blip.tv/chemteam/boyle-s-law-i-the-pressure-volume-relationship-2883136
http://www.grc.nasa.gov/WWW/k-12/airplane/aboyle.html
Below are a video and an animation for Boyle's Law:
http://blip.tv/chemteam/boyle-s-law-i-the-pressure-volume-relationship-2883136
http://www.grc.nasa.gov/WWW/k-12/airplane/aboyle.html
Charles' Law
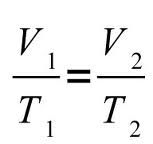
This law was established by Jacques Charles. He studied the interaction between Volume and Temperature. These are directly related, meaning as one increases the other will also increase, and when one decreases, the other will as well.
Here's a video and practice link for Charles' Law:
http://www.youtube.com/watch?v=FQ_m4A0YFpc
http://science.widener.edu/svb/tutorial/charleslawcsn7.html
Here's a video and practice link for Charles' Law:
http://www.youtube.com/watch?v=FQ_m4A0YFpc
http://science.widener.edu/svb/tutorial/charleslawcsn7.html
Gay-Lussac's Law

This law was established by Joseph Louis Gay-Lussac. This chemist studied the relationship between pressure and temperature. What he found was that pressure and temperature are directly related. As temperature increases, pressure will also, and when temperature decreases, so will pressure.
Below is a practice link and educational video for Gay-Lussac's law:
http://www.youtube.com/watch?v=w3kWaDWxChM
http://www.chemteam.info/GasLaw/WS-Gay-Lussac.html
Below is a practice link and educational video for Gay-Lussac's law:
http://www.youtube.com/watch?v=w3kWaDWxChM
http://www.chemteam.info/GasLaw/WS-Gay-Lussac.html
Combined Gas Law

The combined gas law is used when the number of molecules in an equation stays constant. To solve this kind of equation, you will need to just plug in the numbers for the correct variables, which can be seen in the visual to the left. Note: Temperature should always be expressed in Kelvin. (K)
Here is a video to enhance your understanding:
http://www.brightstorm.com/science/chemistry/kinetic-molecular-theory/combined-gas-law/
Here is a video to enhance your understanding:
http://www.brightstorm.com/science/chemistry/kinetic-molecular-theory/combined-gas-law/
Ideal Gas Law
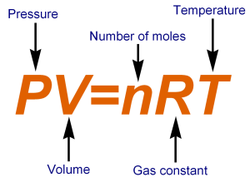
In order to solve a problem using the Ideal Gas Law, you need to know the "R" value, which is the universal gas constant. This value needs to correlate with whatever pressure you are solving with in a given equation. The "R" value can be solved for by dividing (pressure*volume) by (#of moles*temperature) --> (PV/nT)
Here is a link to a video on the ideal gas law:
http://www.brightstorm.com/science/chemistry/kinetic-molecular-theory/ideal-gas-law/
Here is a link to a video on the ideal gas law:
http://www.brightstorm.com/science/chemistry/kinetic-molecular-theory/ideal-gas-law/
Avogadro's Law

This law is used to describe the relationship between volume and amount of gas. The volume of gas is directly proportional to the # of moles of the gas when the temperature and pressure remain constant. Here is a helpful video:
http://www.brightstorm.com/science/chemistry/kinetic-molecular-theory/avogadros-principle/
http://www.brightstorm.com/science/chemistry/kinetic-molecular-theory/avogadros-principle/
Dalton's Law

Dalton's law simply states that the combined partial pressures of each gas in a solution is the sum of the total solution.
Here is a video: http://www.youtube.com/watch?v=tTGehRESHmA
Here is a video: http://www.youtube.com/watch?v=tTGehRESHmA
Diffusion/Effusion Rate
The rate of diffusion/effusion is dependent on mass and size of the hole.
*The heavier the gas particle, the slower it will effuse.
*The lighter the gas particle, the faster it will effuse.
This is solved by dividing the molar mass of the larger substance by the molar mass of the smaller object.
*The heavier the gas particle, the slower it will effuse.
*The lighter the gas particle, the faster it will effuse.
This is solved by dividing the molar mass of the larger substance by the molar mass of the smaller object.
Density

Density also plays a role in gas laws. Molar mass can be determined by the density of gas. It doesn't play a large role in gas laws, however, it is helpful to understand.
Here is a helpful video incorporating the ideal gas law into the density equation:
http://www.youtube.com/watch?v=AHY8yvrkQxc
Here is a helpful video incorporating the ideal gas law into the density equation:
http://www.youtube.com/watch?v=AHY8yvrkQxc